The Science
Low Energy
Emulsification
01 — Foundations
What is an Emulsion?
An emulsion is a heterogeneous system consisting of at least two immiscible liquid phases — most commonly oil and water — where one phase (the dispersed phase) is distributed as fine droplets within the other (the continuous phase). Emulsions are stabilized by surface-active agents, or surfactants, which reduce the interfacial tension between the two phases and form a protective film around each droplet.
Emulsions are ubiquitous in daily life: lotions, creams, salad dressings, mayonnaise, milk, and pharmaceutical suspensions are all emulsions. The quality of an emulsion — its stability, texture, appearance, and efficacy — is fundamentally determined by the size and uniformity of its droplets.
Conventionally, emulsions are produced by applying large amounts of mechanical energy through high-shear mixers, high-pressure homogenizers, or ultrasonicators to break large droplets into smaller ones. This is energy-intensive, time-consuming, and can damage heat-sensitive ingredients. Low Energy Emulsification offers a fundamentally different approach.
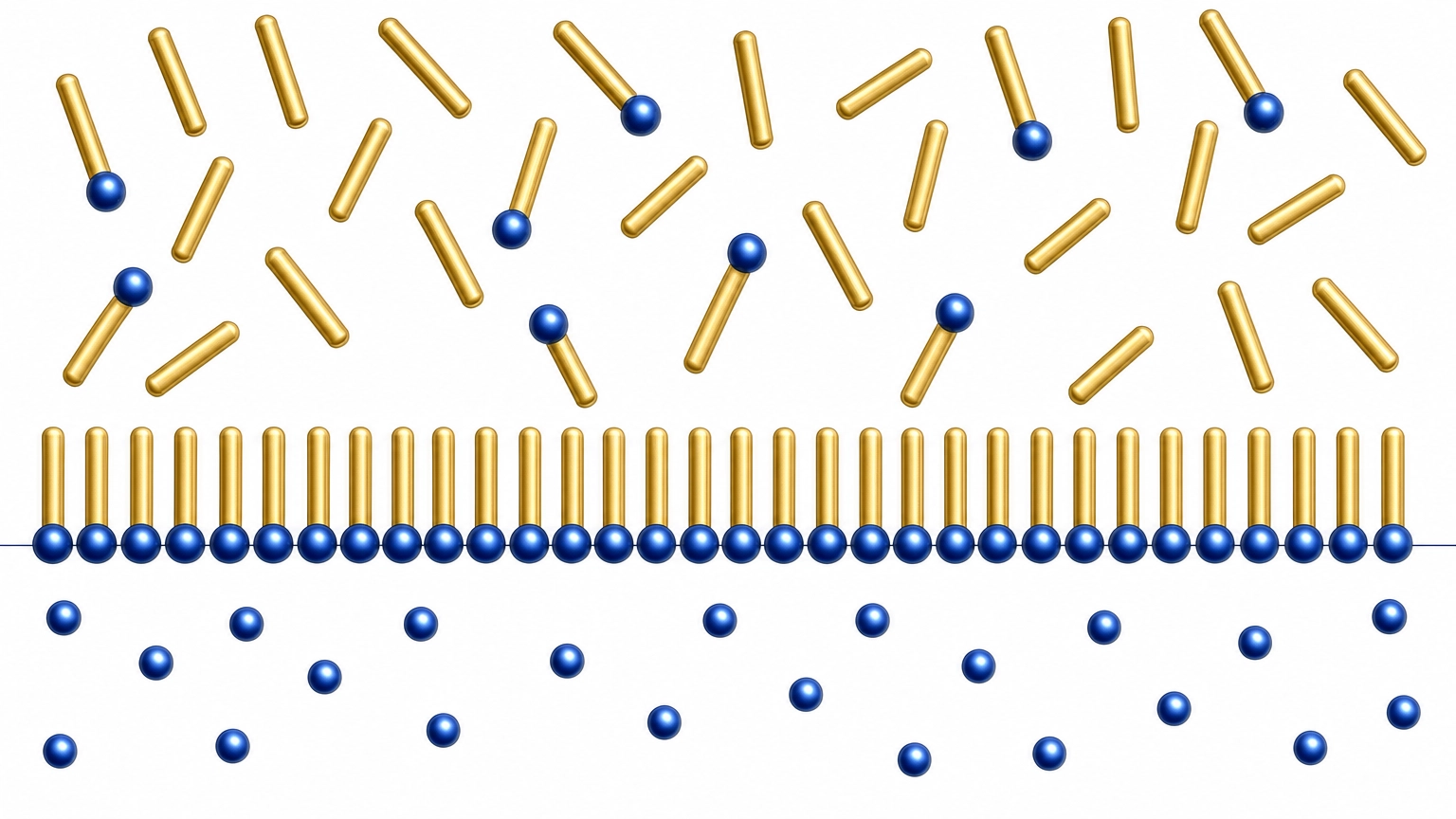
Fig. 1 — Surfactant molecules (gold tails = hydrophobic, blue heads = hydrophilic) self-assembling at the oil-water interface. This thermodynamic self-organization is the basis of LEE.
02 — Core Principles
The Three Laws of LEE
Dr. Lin distilled the philosophy of Low Energy Emulsification into three governing principles that apply to both thermal and mechanical energy in any manufacturing context.
Where Needed
Apply energy only to the specific portion of the formulation that requires it. In practice, this means heating only the β phase (a small fraction of the total batch) rather than the entire contents of the compounding kettle.
When Needed
Apply energy at the precise moment in the manufacturing sequence where it has the greatest effect. Thermal energy applied at the wrong stage is wasted; applied at the inversion point, it is transformative.
In the Amount Needed
Minimize the total energy input by optimizing the ratio of heated to unheated phases. By maximizing the ratio of the cold α phase to the hot β phase, the total heat removed — and therefore the cooling time — is dramatically reduced.
03 — Methods
Primary LEE Techniques
Self-Emulsification
Spontaneous Emulsification
When two immiscible liquids are brought into contact under non-equilibrium conditions, a chemical potential gradient drives the spontaneous formation of an emulsion. Rapid diffusion of surfactants or solvents across the interface causes local interfacial turbulence and the budding of fine droplets — with no mechanical agitation required.
Applications
- —Self-nano-emulsifying drug delivery (SNEDDS)
- —Essential oil dispersions
- —Flavor encapsulation
PIT Method
Phase Inversion Temperature
Nonionic surfactants (such as ethoxylated compounds) change their preferred curvature with temperature. At low temperatures they favor oil-in-water (O/W) emulsions; at high temperatures, water-in-oil (W/O). By heating the system to the Phase Inversion Temperature (PIT) and then rapidly cooling it, ultra-fine droplets are trapped in a kinetically stable state.
Applications
- —Cosmetic creams and lotions
- —Pharmaceutical topical formulations
- —Food emulsions
PIC Method
Phase Inversion Composition
At constant temperature, water is slowly titrated into an oil-surfactant mixture (or vice versa). As the phase ratio shifts, the preferred curvature of the surfactant film changes, inducing a phase inversion. At the inversion point, interfacial tension drops to near zero, allowing extremely fine droplets to form under gentle stirring alone.
Applications
- —Room-temperature formulation of heat-sensitive actives
- —Vitamin and antioxidant encapsulation
- —Fragrance emulsions
High Internal Phase Emulsions
Gel Emulsification
In gel-emulsions, the dispersed phase volume fraction can exceed 0.74, creating a system with gel-like rheology. The immense internal friction during simple mixing effectively breaks down droplets without external high-shear devices. These systems are particularly advantageous for emulsifying highly viscous oils.
Applications
- —Viscous oil emulsification
- —Concentrated cosmetic formulas
- —Industrial lubricant emulsions
04 — Advantages
Why LEE Matters
Energy Savings
Reduction in total manufacturing energy consumption compared to conventional hot-process methods.
Productivity Gain
Increase in batch throughput without capital investment in new equipment, by dramatically shortening heating and cooling cycles.
Droplet Quality
Phase inversion methods produce nanoemulsions with droplet sizes and size distributions that match or exceed high-energy methods.
Ingredient Protection
Room-temperature methods protect heat-sensitive vitamins, essential oils, and pharmaceutical actives from thermal degradation.